0 引 言
1 阴极催化层微结构重建
Table 1 Composition of the catalyst layer表 1 催化层的各相组成 |
| Component | Phase | Volume fraction / % |
|---|---|---|
| Carbon black | MS | 34.72 |
| Pt | MS | 1.24 |
| Ionomer | MS | 24.04 |
| Pore | P | 40.00 |
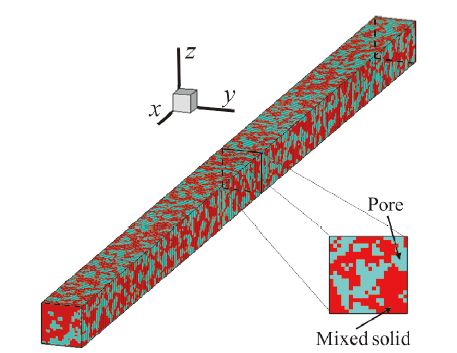
Fig. 1 SEM micrograph of cathode catalyst layer图1 阴极催化层的SEM图 |
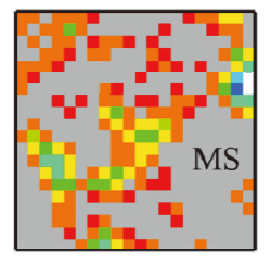
Fig. 2 Result from the SGM reconstruction (red for mixed solid and light blue for the pore)图 2 重建结果(红色为固体混合相,淡蓝色为孔相) |
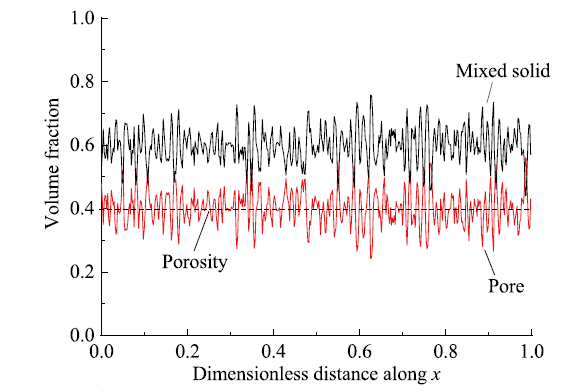
Fig. 3 Evolution of volume fraction of each phase along the direction of electrode thickness图3 各相体积分数沿电极厚度方向的变化 |
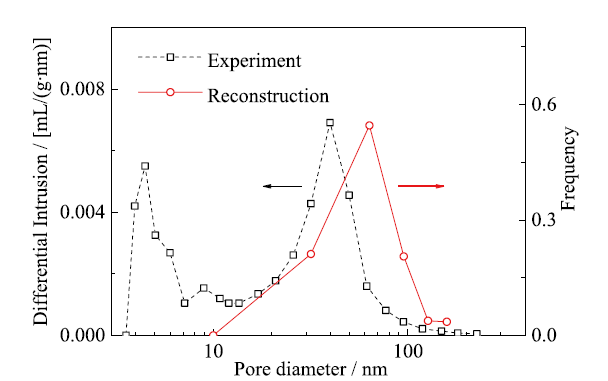
Fig. 4 Pore diameter distribution for the reconstructed CL and compared with the experimental result图4 重建催化层的孔径分布与实验结果的对比 |
2 数值模型
2.1 模型假设
Fig. 5 Schematic of charge and species transfer in cathode catalyst layer图5 阴极催化层模型的电荷及物质输运示意图 |
2.2 控制方程
Table 2 Governing equations and source terms of the model表2 模型主控方程及源项 |
| Categories | Governing equations | Source terms | |
|---|---|---|---|
| P | MS | ||
| Species | $\frac{\partial \left( \varepsilon {{C}^{{{\text{O}}_{\text{2}}}}} \right)}{\partial t}=\nabla \cdot \left( D_{\text{eff}}^{{{\text{O}}_{\text{2}}}}\nabla {{C}^{{{\text{O}}_{\text{2}}}}} \right)+S_{\text{c}}^{{{\text{O}}_{\text{2}}}}$ | $S_{\text{c}}^{{{\text{O}}_{\text{2}}}}=-\frac{{{s}^{{{\text{O}}_{\text{2}}}}}{{j}_{\text{c}}}}{nF}$ | / |
| $\frac{\partial \left( \varepsilon {{C}^{{{\text{H}}_{\text{2}}}\text{O}}} \right)}{\partial t}=\nabla \cdot \left( D_{\text{eff}}^{{{\text{H}}_{\text{2}}}\text{O}}\nabla {{C}^{{{\text{H}}_{\text{2}}}\text{O}}} \right)+S_{\text{c}}^{{{\text{H}}_{\text{2}}}\text{O}}$ | $S_{\text{c}}^{{{\text{H}}_{\text{2}}}\text{O}}=-\frac{{{s}^{{{\text{H}}_{\text{2}}}\text{O}}}{{j}_{\text{c}}}}{nF}-{{S}_{\text{ice}}}\text{+}{{S}_{\text{balance}}}$ | $S_{\text{c}}^{{{\text{H}}_{\text{2}}}\text{O}}=-\nabla \cdot \left( \frac{{{n}_{\text{d}}}}{F}{{\mathrm{i}}_{\text{e}}} \right)-{{S}_{\text{balance}}}$ | |
| Charge | $0=\nabla \cdot \left( \kappa _{\text{eff}}^{\text{e}}\nabla {{\phi }_{\text{e}}} \right)+{{S}_{\phi ,\text{e}}}$ | / | ${{S}_{\phi ,\text{e}}}={{j}_{\text{c}}}$ |
| $0=\nabla \cdot \left( \sigma \nabla {{\phi }_{\text{s}}} \right)+{{S}_{\phi ,\text{s}}}$ | / | ${{S}_{\phi ,\text{s}}}=-{{j}_{\text{c}}}$ | |
Table 3 Physical properties and other parameters of the material表3 材料的物性及部分计算参数 |
| Parameters | Value | Units |
|---|---|---|
| Cathode exchange current density i0,ref[2] | 104 | A/m3 |
| Cathode equilibrium potential U0[2] | U0=1.23-9.0×10-4(T-298.15) | V |
| Electro-osmotic drag coefficient nd[21] | ${{n}_{\text{d}}}={{a}_{T}}\left\{ \begin{align} & 0\text{ }\lambda \le \text{3} \\ & {{\left( \frac{\lambda -3}{3} \right)}^{4}}\text{ 3}\lambda \le \text{6} \\ & 1\text{ others} \\ \end{align} \right.$${{a}_{T}}=\left( -\frac{1484.21}{{{T}_{\text{a}}}}+6.062974 \right)\text{ ; if }{{a}_{T}}\text{0}\text{.2, }{{a}_{T}}\text{=0}\text{.2 }$ | H2O/H+ |
| Cathode transfer coefficient αc[2] | 1 | |
| Reference oxygen concentration $C_{\text{ref}}^{{{\text{O}}_{\text{2}}}}$[2] | 40 | mol/m3 |
| Ambient temperature Ta | 243.15(set as default)/248.15/253.15/258.15 | K |
| Current loading | 2 000/3 000(set as default)/4 000 | A/m2 |
| Diffusion coefficient of oxygen/vapor $D_{\text{b}}^{i}$[20] | 1.327 × 10-7/1.28 × 10-5 | m2/s |
| Bruggeman coefficient b[2] | 1.5(set as default)/2.5 | |
| Faraday constant F | 964 87 | C/mol |
| Universal gas constant R | 8.314 | J/(mol·K) |
| Conductivity of carbon black σ[20] | 104 | S/m |
| *Equivalent weight of ionomer EW | 1.0 | kg/mol |
| *Volume fraction of ionomer at mix solid phase ${{\varepsilon }_{\text{m}}}$ | 0.400 7 | |
| Net-water transport coefficient α[22] | -0.1/-0.2(set as default)/-0.4/-0.6 | |
| Density of O2/N2/ice/vapor/carbon/ionomer ρ[2] | 1.4/1.2/0.6/1 800/1 820 | kg/m3 |
| **Specific surface area of phase interface $A_{\text{inter}}^{{}}/\text{V}$ | 2.67 × 1011 | 1/m |
| *Specific electro-chemical active area $A_{\text{inter}}^{\text{r}}/\text{V}$ | 1.064 × 1011 | 1/m |
| *Average pore size $\overline{d}$ | 40 | nm |
| *Porosity of the GDL ${{\varepsilon }_{\text{GDL}}}$ | 0.513 | |
| *Thickness of the GDL ΔX | 200 | μm |
Notes: * data from collaborators; ** results from the reconstructed structure. |
2.3 初边界条件和求解方法
3 结果与分析
Table 4 Cases summary表4 算例汇总 |
| Series | α | $\frac{I}{{{A}_{\text{s}}}}$/ (A/m2) | Ta / K | Others |
|---|---|---|---|---|
| Base case | -0.2 | 3 000 | 243.15 | $g({{{\varepsilon }'}_{\text{ice}}})=1-{{{\varepsilon }'}_{\text{ice}}}$; b = 1.5 |
| Case 1 | -0.1/-0.4/-0.6 | 3 000 | 243.15 | $g({{{\varepsilon }'}_{\text{ice}}})=1-{{{\varepsilon }'}_{\text{ice}}}$; b = 1.5 |
| Case 2 | -0.2 | 2 000/4 000 | 243.15 | $g({{{\varepsilon }'}_{\text{ice}}})=1-{{{\varepsilon }'}_{\text{ice}}}$.; b = 1.5 |
| Case 3 | -0.2 | 3 000 | 258.15/253.15/248.15 | $g({{{\varepsilon }'}_{\text{ice}}})=1-{{{\varepsilon }'}_{\text{ice}}}$; b = 1.5 |
| Case 4 | -0.2 | 3 000 | 243.15 | $g({{{\varepsilon }'}_{\text{ice}}})={{\left( 1-{{{{\varepsilon }'}}_{\text{ice}}} \right)}^{2}}$; b = 1.5 $g ({{{\varepsilon }'}_{\text{ice}}})=1-{{{\varepsilon }'}_{\text{ice}}}$; b = 2.5 |
3.1 结冰形貌(基础算例)
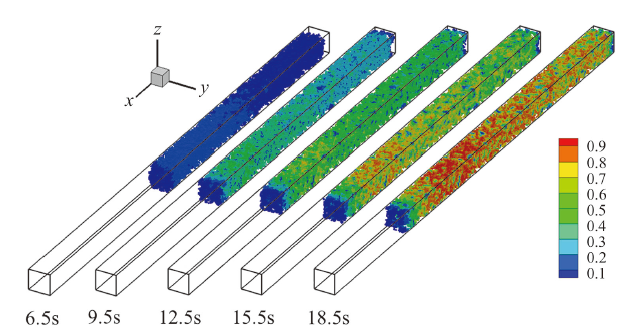
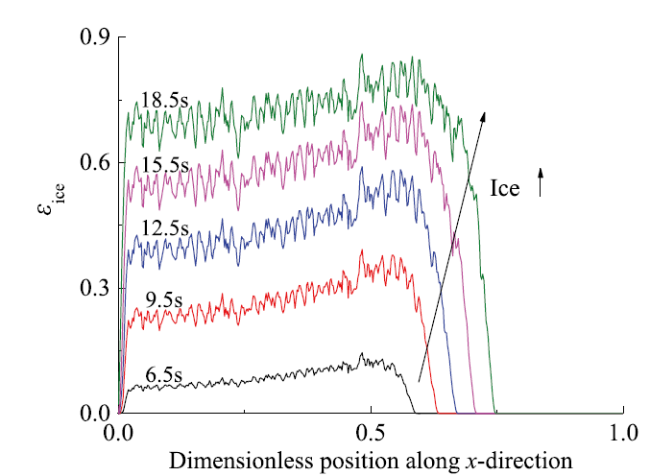
Fig. 6 Ice formation in CL and its time-change图6 催化层内含冰量分布及其随时间演化 |
Fig. 7 Ice distribution on the central x-sectional-plane at 18.5 s图7 t = 18.5 s时催化层中部x截面的含冰量分布 |
Fig. 8 Ice fraction profile along the x-axis and its time-change图8 沿x轴方向的含冰量分布及其随时间演化 |
3.2 结冰对传质和电化学反应的影响(基础算例)
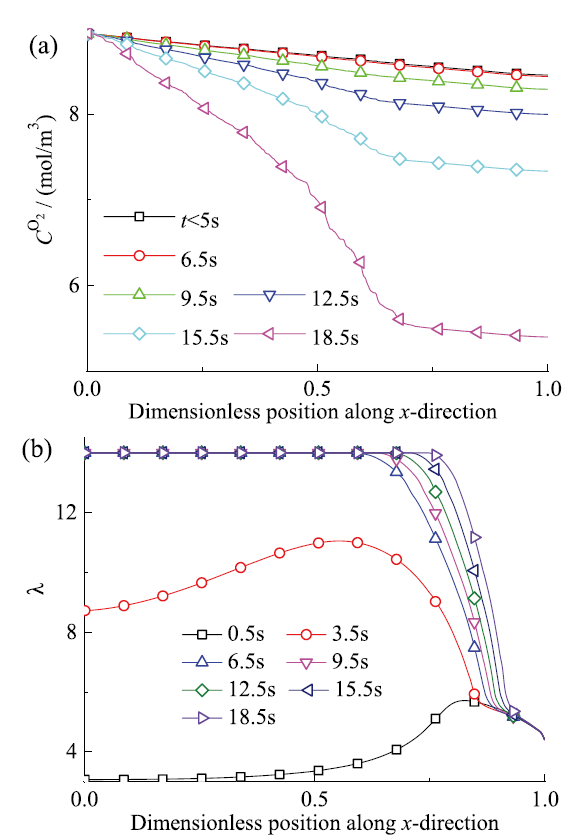
Fig. 9 Distribution of mean oxygen concentration (a) and water content (b) along the x-axis and their evolution over time图9 氧气平均浓度(a)和含水量(b)在沿x轴方向的分布及其随时间演化 |
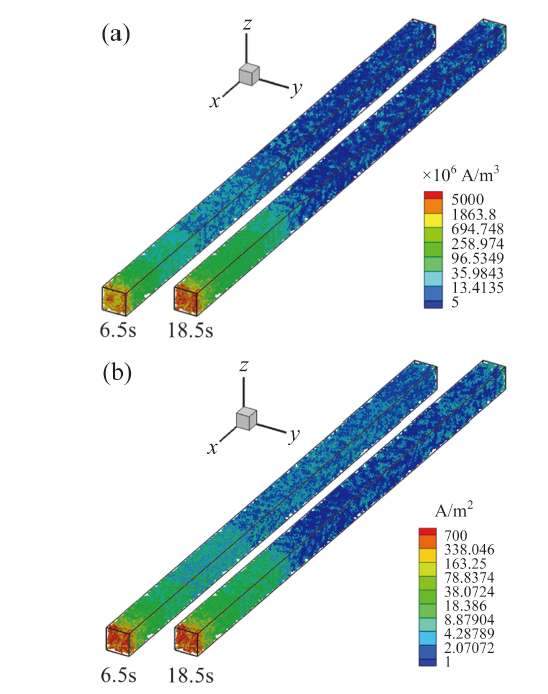
Fig. 10 Distribution of the transfer current density (a) and protonic current density (b) in CL图10 催化层内输运电流密度(a)与质子电流密度(b)分布 |
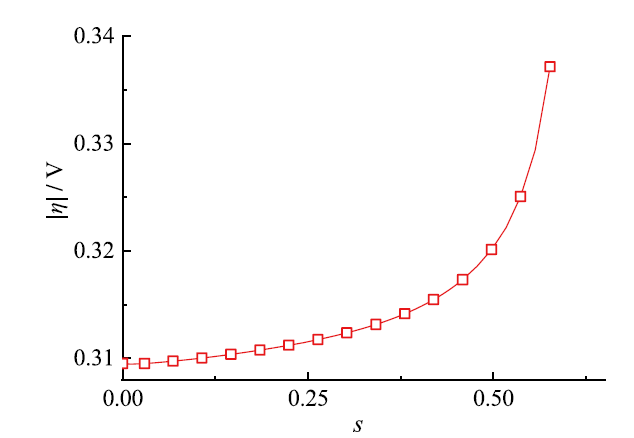
Fig. 11 Evolution of the average overpotential with ice fraction图11 平均过电势随含冰量的演化 |
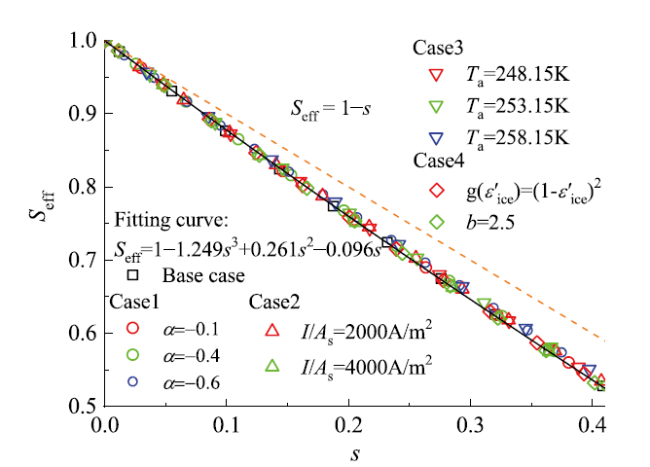
3.3 电化学有效反应面积与结冰量的关系式
Fig. 12 Plots for different conditions of total ice fraction to ECA图12 不同条件下电化学有效反应面积与总含冰量的关系 |