0 引言
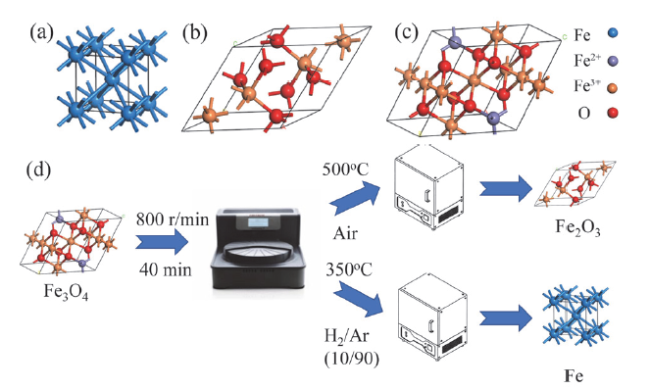
Fig. 1 The crystal structures of Fe (a), Fe2O3 (b) and Fe3O4 (c); (d) scheme of catalyst preparation图1 Fe(a)、Fe2O3(b)和Fe3O4(c)的晶体结构示意图;(d)材料制备流程示意图 |
1 实验部分
1.1 试剂与仪器
1.2 催化剂的制备
1.3 实验准备
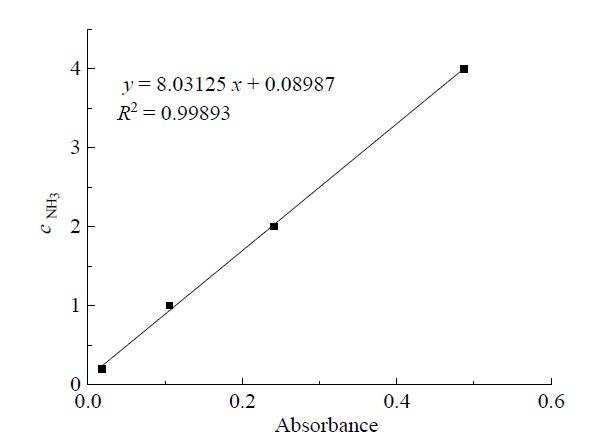
Fig. 2 Standard curve of NH3/NH4+ concentration图2 NH3/NH4+ 摩尔浓度的标准曲线 |
1.4 电化学实验
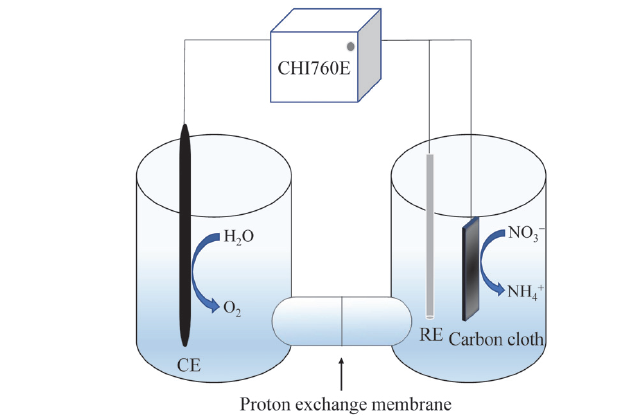
Fig. 3 Diagram of electrolytic cell图3 电解池示意图 |
1.5 经济分析中的成本计算
2 结果与讨论
2.1 硝酸根的还原反应
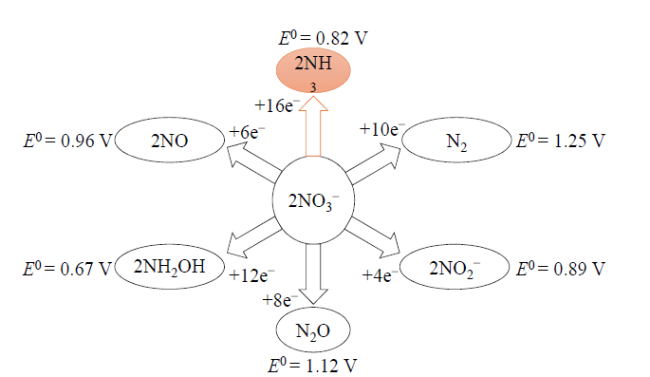
Fig. 4 Reduction products of nitrate图4 硝酸盐的还原产物 |
2.2 单质Fe、Fe2O3和Fe3O4的表征
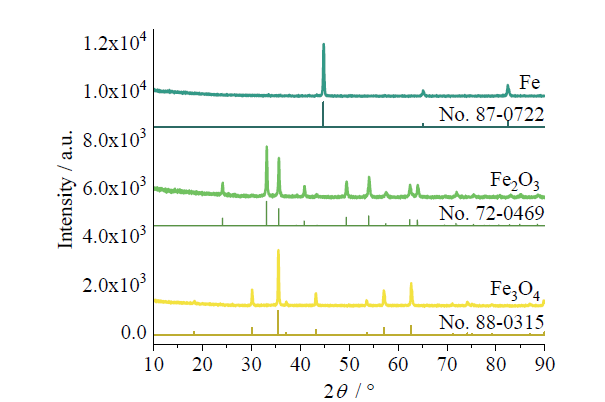
Fig. 5 XRD patterns of Fe, Fe2O3, and Fe3O4图5 单质Fe、Fe2O3和Fe3O4的XRD图 |
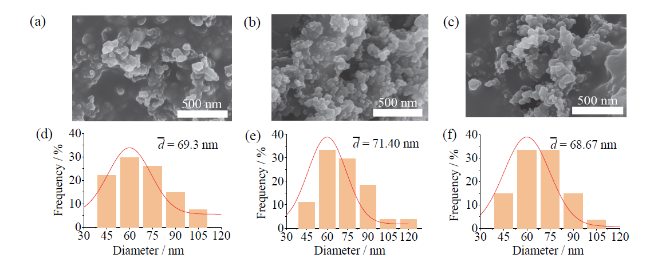
Fig. 6 The corresponding SEM images (a-c) and particle size distribution (d-f) of Fe, Fe2O3 and Fe3O4 samples, respectively图6 单质Fe、Fe2O3和Fe3O4样品对应的SEM图片(a-c)和粒径分布图(d-f) |
2.3 电催化NARR性能测试
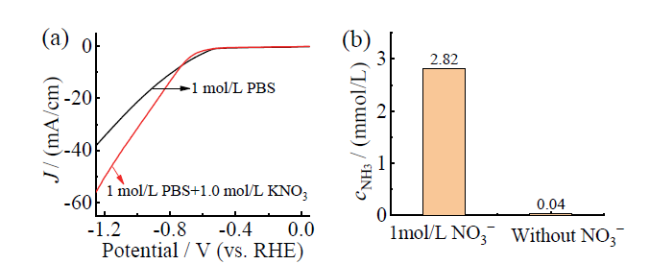
Fig. 7 Comparison of electrochemical properties of Fe3O4 in buffer solutions with and without KNO3: (a) LSV; (b) cNH3图7 Fe3O4在含有KNO3缓冲溶液和不含KNO3的缓冲溶液中的电化学性能对比:(a)LSV;(b)cNH3 |
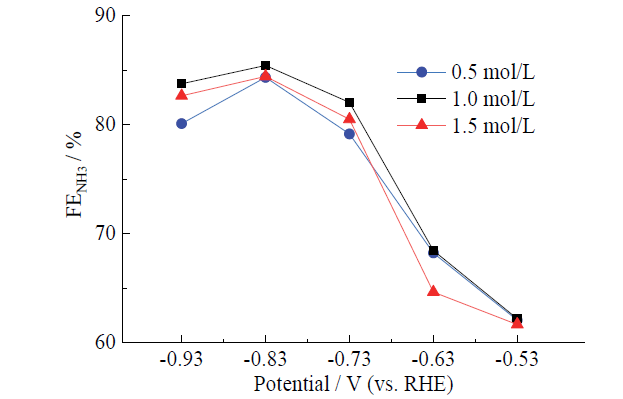
Fig. 8 Comparison of Faraday efficiency of Fe3O4 in different concentrations of phosphate buffer solutions图8 Fe3O4在不同浓度磷酸缓冲溶液中的法拉第效率对比 |
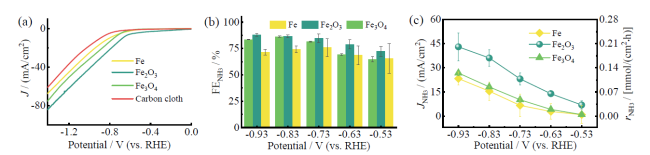
Fig. 9 Comparison of NARR performance in 1.0 mol/L NO3- by three iron-based catalysts: (a) LSV curves; (b) FENH3; (c) JNH3 as well as the corresponding rNH3图9 三种铁系催化剂在1.0 mol/L NO3- 电解液中的NARR性能对比:(a)LSV;(b)FENH3;(c)JNH3与对应的rNH3 |
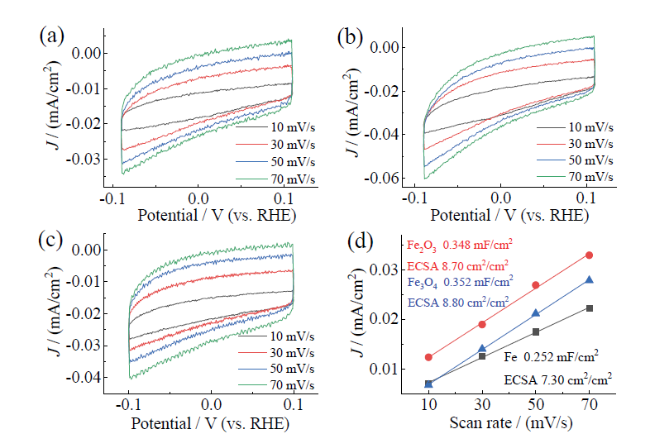
Fig. 10 Electrochemical active surface area: Fe (a), Fe2O3 (b) and Fe3O4 (c) with various scan rates from 10 mV/s to 70 mV/s; (d) plots of the current density versus the scan rate for Fe, Fe2O3 and Fe3O4图10 电化学活性面积:Fe(a)、Fe2O3(b)、Fe3O4(c)在10 ~ 70 mV/s的CV曲线;(d)电流密度和扫描速度的关系 |
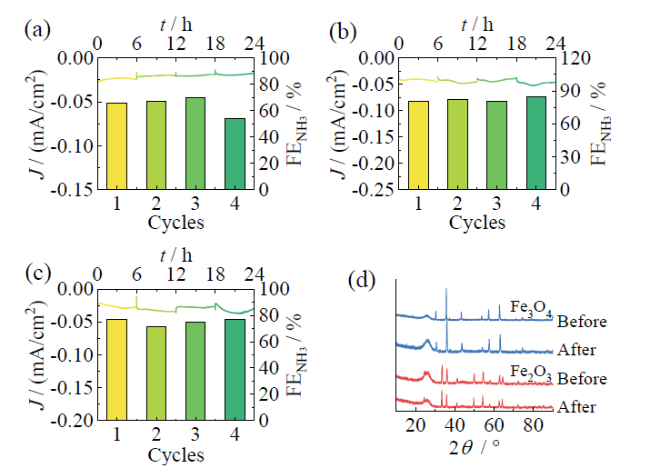
Fig. 11 The J-t curves and the corresponding FENH3 per cycle of Fe (a), Fe2O3 (b) and Fe3O4 (c) at -0.83 V; (d) comparison of XRD patterns for Fe3O4 and Fe2O3 before and after the reaction图11 Fe(a)、Fe2O3(b)和Fe3O4(c)在 -0.83 V电压下J-t曲线以及每次循环的FENH3;(d)Fe3O4和Fe2O3在反应前后XRD对比 |
2.4 技术经济分析
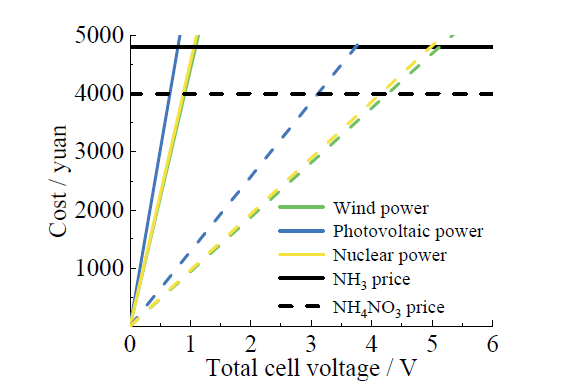
Fig. 12 Economic analysis of electrocatalytic nitrate reduction products based on new energy图12 基于新能源的电催化硝酸盐还原产品的经济性分析 |