0 引言
1 锂离子电池快充性能提高方法
1.1 材料的选择
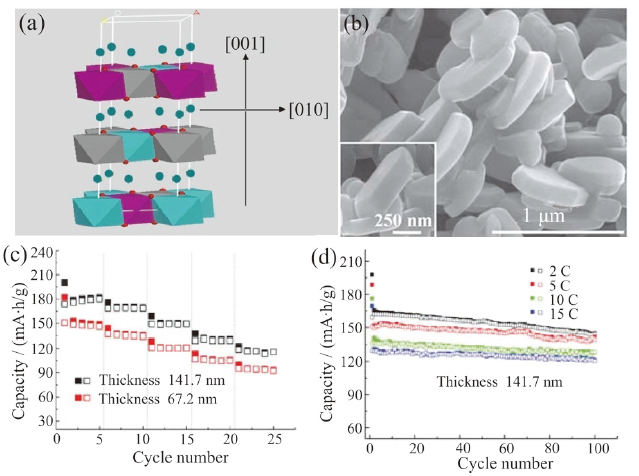
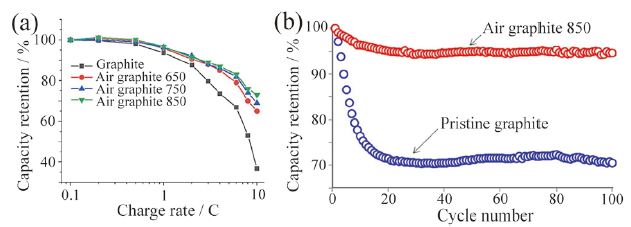
Fig. 2 (a) Crystal structure of LiNi1/3Mn1/3Co1/3O2 (the red and green spheres are oxygen and lithium atoms, respectively; the octahedra represents MO6 unit); (b) SEM diagram of synthesized LiNi1/3Mn1/3Co1/3O2 material; (c) rate performances of the materials with different thickness; (d) cycling performances of material with 141.7 nm thickness at high rates[37]图2 (a)LiNi1/3Mn1/3Co1/3O2材料晶体结构示意图(红色和绿色球分别代表氧原子和锂原子,八面体代表MO6基本单元);(b)LiNi1/3Mn1/3Co1/3O2材料SEM图;(c)不同厚度材料倍率性能;(d)具有141.7 nm厚度材料在高倍率下的循环性能[37] |
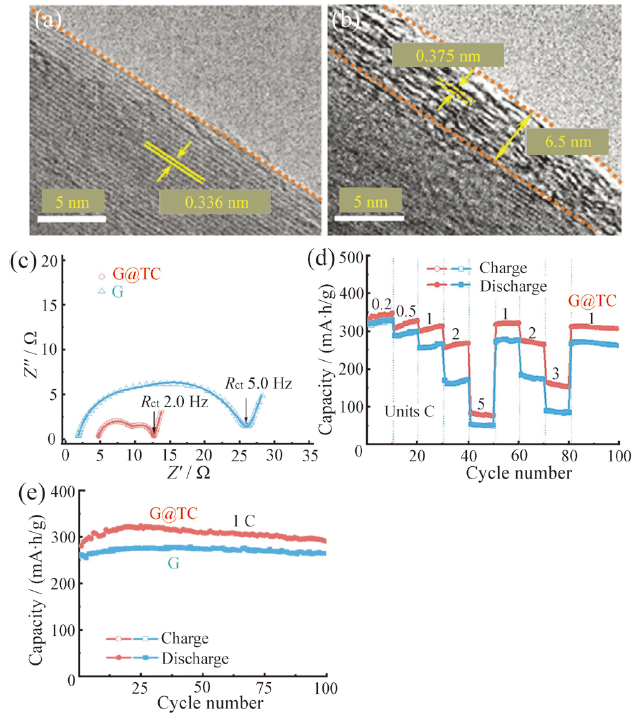
Fig. 3 (a) TEM images of G material; (b) TEM images of G@TC material; (c) electrochemical impedance spectra of G and G@TC material; (d) rate performance of G and G@TC material; (e) cycling performance of G and G@TC material at a current density of 1 C[49]图3 (a)材料G的TEM图;(b)材料G@TC 的TEM图;(c)材料G和材料G@TC的阻抗图;(d)材料G和材料G@TC的倍率性能曲线;(e)材料G和材料G@TC在1 C倍率下循环性能曲线[49] |
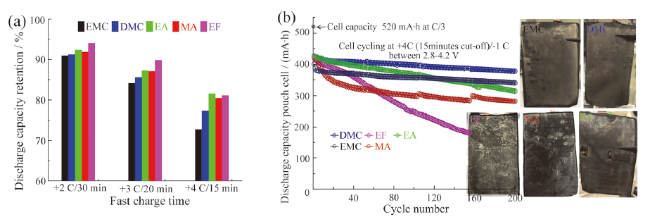
Fig. 7 (a) Discharge capacity retention diagrams of different solvents under different rates; (b) long-term cycling performance of the pouch cell under fast charging conditions with different electrolytes and Li plating diagrams of graphite electrodes[66]图7 (a)不同溶剂在不同充电倍率下放电容量保持率图;(b)不同溶剂在4 C倍率下长期循环性能图及负极界面析锂[66] |
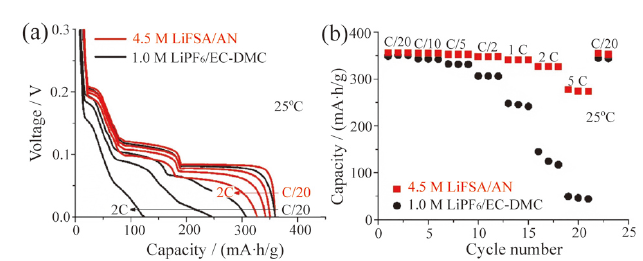
Fig. 10 (a) First charge and discharge voltage profiles of NCM622/graphite full cells at a C/10 charging rate before cycling test; (b) Nyquist plots of the NCM622/graphite full cells at a charged state after 300 cycles with two electrolytes; (c) cycling performance plots of the NCM622/graphite full cells with two electrolytes[73]图10 (a)两种电解液的NCM622/石墨电池循环以C/10充电倍率下首次充放电曲线图;(b)两种电解液的NCM622/石墨电池循环300次后的阻抗图;(c)在两种电解液中NCM622/石墨电池循环性能图[73] |
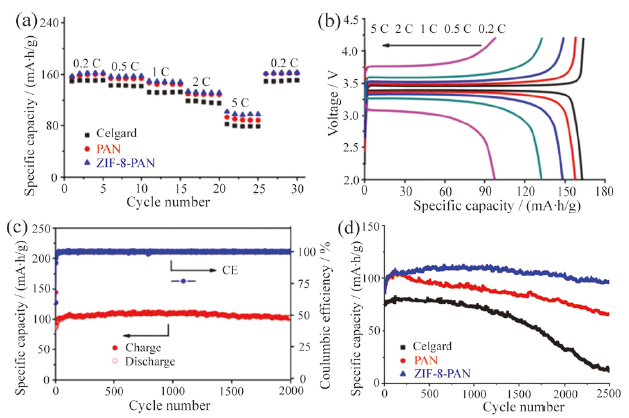
Fig. 11 Electrochemical performance of LiFePO4-Li cells with the ZIF-8/PAN composite fibrous separator: (a) rate performance curves; (b) rate charge-discharge curves; (c) cycle performance of ZIF-8/PAN composite fibrous separator with 5 C current density; (d) cycle performance of three separators cell with 5 C current ZIF-8/PAN composite fibrous[79]图11 ZIF-8/PAN 复合纤维隔膜的LiFePO4-Li电池电化学性能:(a)倍率曲线图;(b)充放电倍率曲线图;(c)在5 C 电流下复合隔膜电池长期循环性能图;(d)在5 C 电流下三种隔膜电池长期循环性能图[79] |
1.2 电极设计的合理选择
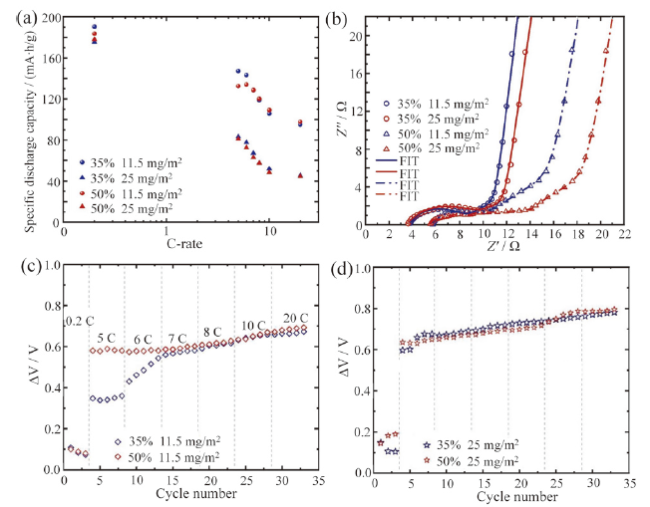
Fig. 12 Specific discharge capacity (a) and Nyquist plots (b) of NCM622 electrode with different porosity at different area density; polarization diagrams of the NCM622 electrode with different porosity under various rates at 11.5 mg/cm2 (c) and 25 mg/cm2 (d) area density[83]图12 具有两种不同面密度和不同孔隙率的NCM622电极倍率性能(a)和阻抗图(b);具有11.5 mg/cm2(c)和25 mg/cm2(d)面密度的电极在不同倍率和不同孔隙率下电极极化图[83] |
1.3 充电策略的合理选择
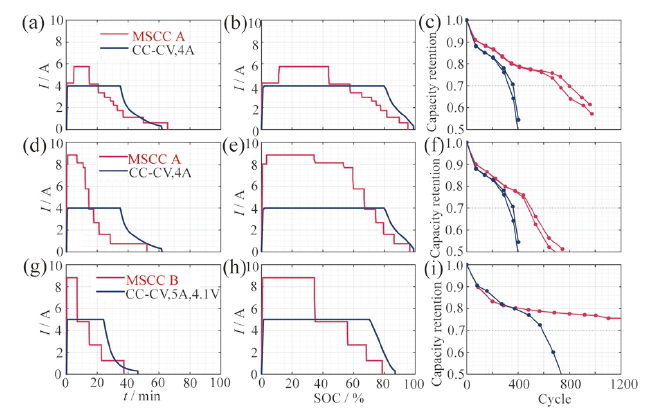
Fig. 13 Different charge protocols results: (a,d,g) curve of charge current as a function of time; (b,e,h) curve of charge current as a function of SOC; (c,f,i) curve of capacity retention as a function of cycle number[88]图13 不同充电策略性能结果:(a、d、g)充电电流与时间关系曲线;(b、e、h)充电电流与电池SOC关系曲线;(c、f、i)循环次数与容量保持率关系曲线[88] |
| 组号 | 充电电流 / C | 充电时间 / min | ||
|---|---|---|---|---|
| SOC = 0% ~ 30% | SOC = 30% ~ 60% | SOC = 60% ~ 80% | ||
| 1 | 2.2 | 1.9 | 0.9 | 31.0 |
| 2 | 2.2 | 1.9 | 0.7 | 34.8 |
| 3 | 2.2 | 1.7 | 0.9 | 32.1 |
| 4 | 2.2 | 1.7 | 0.7 | 35.9 |
| 5 | 2.2 | 1.5 | 0.9 | 33.5 |
| 6 | 2.0 | 1.9 | 0.9 | 31.8 |
| 7 | 2.0 | 1.9 | 0.7 | 35.6 |
| 8 | 2.0 | 1.7 | 0.9 | 32.9 |
| 9 | 2.0 | 1.5 | 0.9 | 34.3 |
| 10 | 1.8 | 1.9 | 0.9 | 32.8 |
| 11 | 1.8 | 1.7 | 0.9 | 33.9 |
| 12 | 1.8 | 1.5 | 0.9 | 35.3 |
| 13 | 1.5 | 1.5 | 1.5 | 32.0 |
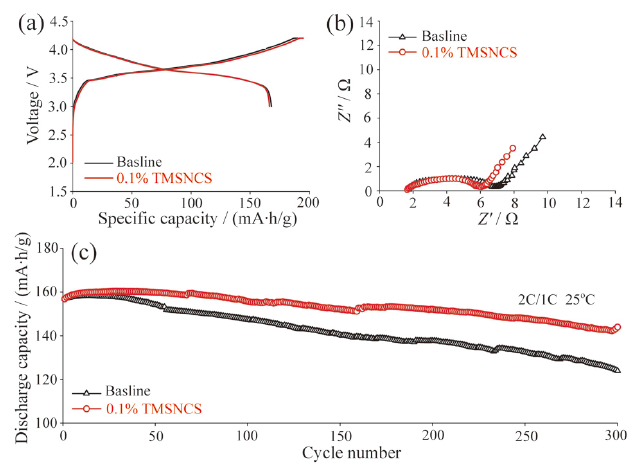
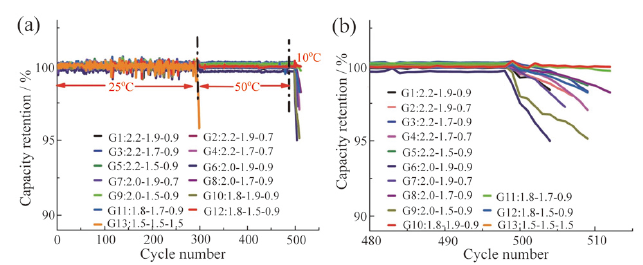
Fig. 16 (a) Time derivative curve of voltage at different average charge temperatures; (b) capacity retention of the four cells during the fast charge cycling test at different charge temperatures; (c-f) photos of the graphite anodes from cells after the fast charge cycling test at different charge temperatures[90]图16 (a)不同充电温度下电池时间电压微分曲线;(b)不同充电温度下快充电芯容量保持率;(c ~ f)不同充电温度下快充电芯负极界面图[90] |