0 引言
1 全无机钙钛矿太阳能电池结构
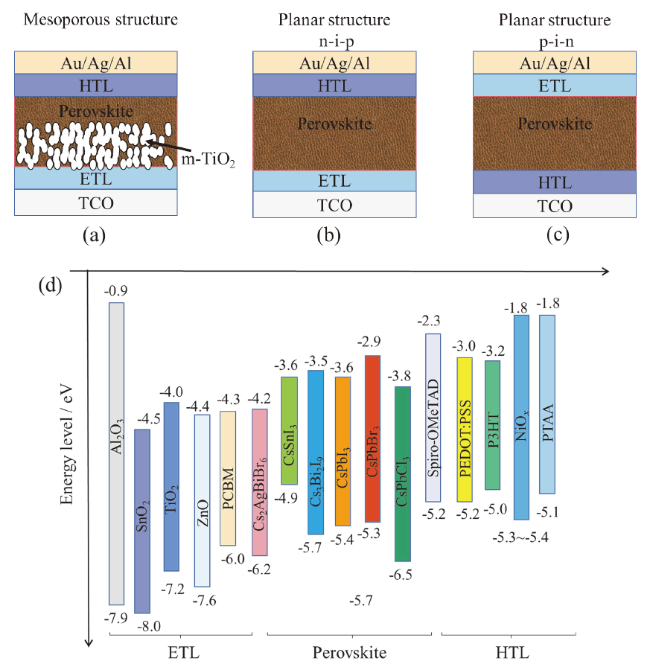
Fig. 1 (a) TiO2 mesoporous structure; (b) planar normal-structure; (c) planar invers-structure; (d) energy level diagram of each layer of inorganic perovskite solar cell图1 (a)TiO2介孔结构;(b)平面正置结构;(c)平面反置结构;(d)无机钙钛矿太阳能电池中各层材料能级图 |
2 不同结构全无机钙钛矿材料的应用
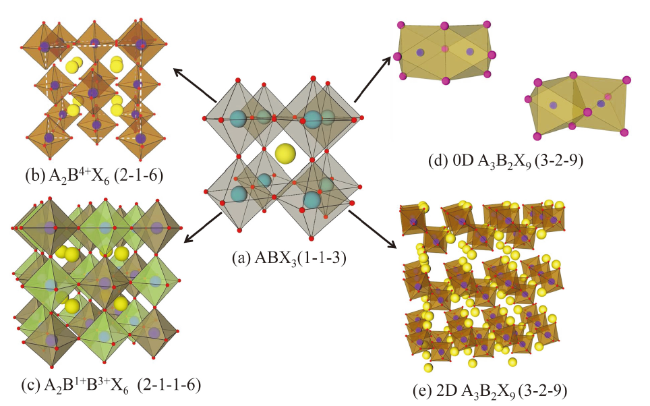
Fig. 2 Crystal structures of different perovskite materials图2 不同钙钛矿材料晶体结构 |
2.1 基于ABX3型钙钛矿的太阳能电池
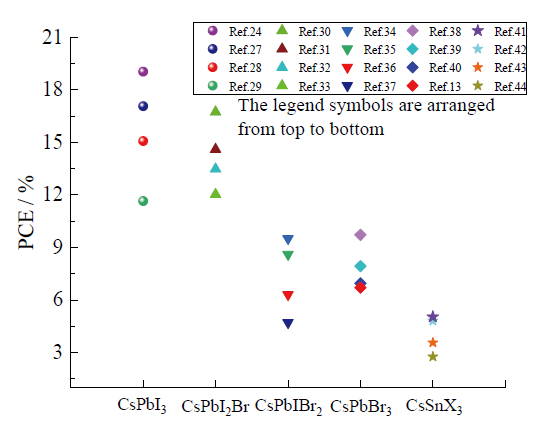
Fig. 3 Efficiency characteristics of ABX3 type inorganic perovskite materials in different device configurations图3 ABX3型无机钙钛矿材料在不同器件构型的效率特性 |
2.2 基于双钙钛矿材料的太阳能电池
Table 1 Efficiency characteristics of A2BX6 and A2B1+B3+X6 inorganic perovskites in different device configurations表1 A2BX6及A2B1+B3+X6型无机钙钛矿材料在不同器件构型中的效率特性 |
| Structure type | Device structure | PCE / % | JSC / (mA/cm2) | VOC / V | FF / % | Ref. |
|---|---|---|---|---|---|---|
| A2BX6 | FTO/TiO2/Cs2SnI6/PEDOT/FTO | 6.10 | 14.10 | 0.62 | 70.0 | [48] |
| A2B1+B3+X6 | FTO/TiO2/Cs2AgBiBr6/Spiro-OMeTAD/Au | 2.43 | 3.93 | 0.98 | 63.0 | [49] |
| FTO/TiO2/Cs2AgBiBr6/PTAA/Au | 1.26 | 1.84 | 1.02 | 67.0 | [50] | |
| FTO/TiO2/Cs2AgBiBr6/P3HT/Au | 1.37 | 1.79 | 1.12 | 68.0 | [51] | |
| ITO/Cu-NiO/Cs2AgBiBr6/C60/BCP/Ag | 2.23 | 3.19 | 1.01 | 69.2 | [52] | |
| ITO/TiO2/Cs2AgBiBr6/C60Spiro-OMeTAD/MoO3/Ag | 2.51 | 3.82 | 1.01 | 65.0 | [53] | |
| ITO/TiO2/Cs2NaBiI6/Spiro-OMeTAD/Au | 0.42 | 1.99 | 0.47 | 44.0 | [54] | |
| FTO/TiO2/Cs2TiBr6/P3HT/Au | 3.28 | 5.69 | 1.02 | 56.4 | [55] |
2.3 基于类钙钛矿材料的太阳能电池
Table 2 The performance and configuration of perovskite solar cells based on A3B2X9 and other typical lead-free perovskite analogs表2 基于A3B2X9及其他无铅类钙钛矿材料的太阳能电池性能及构型 |
| Structure type | Device structure | PCE / % | JSC / (mA/cm2) | VOC / V | FF / % | Ref. |
|---|---|---|---|---|---|---|
| 0D | FTO/TiO2/Cs3Bi2I9/CuI/Ag | 3.20 | 5.78 | 0.86 | 64.38 | [57] |
| FTO/TiO2/Cs3Bi2I9/Spiro-OMeTAD/Ag | 1.77 | 4.45 | 0.79 | 50.34 | [57] | |
| FTO/TiO2/Cs3Bi2I9/PTAA/Ag | 2.30 | 4.82 | 0.83 | 57.49 | [57] | |
| FTO/c-TiO2/m-TiO2/Cs3Bi2I9/Spiro-OMeTAD/Ag | 1.09 | 2.15 | 0.85 | 60.00 | [58] | |
| 2D | ITO/PEDOT:PSS/Cs3Sb2I9/PCBM/Al | 1.49 | 5.31 | 0.72 | 38.97 | [59] |
| ITO/TiO2/Rb3Sb2I9/Spiro-OMeTAD/Au | 1.37 | 4.25 | 0.55 | 59.50 | [60] | |
| ITO/NiOx/Cs3Sb2I6Br3/PCBM/C60/BCP/Ag | 1.15 | 3.15 | 0.64 | 57.00 | [61] | |
| Other materials | FTO/c-TiO2/m-TiO2/AgBiI4/PTAA/Au | 2.20 | 5.24 | 0.67 | 62.09 | [62] |
| FTO/c-TiO2/m-TiO2/Ag2BiI5/PTAA/Au | 2.60 | 6.04 | 0.69 | 62.40 | [62] | |
| FTO/TiO2/Ag3BiI6/PTAA/Au | 4.30 | 10.70 | 0.63 | 64.00 | [63] | |
| FTO/c-TiO2/m-TiO2/AgBi2I7/P3HT/Au | 2.12 | 4.83 | 0.62 | 60.00 | [64] | |
| ITO/CuBiI4/Spiro/Au | 1.11 | 7.18 | 0.38 | 28.67 | [65] | |
| FTO/TiO2/CsBi3I10/Carbon | 1.51 | 4.75 | 0.46 | 69.10 | [66] |