0 引言
1 实验部分
1.1 实验材料
1.2 酸改性凹凸棒石的制备
1.3 酸改性凹凸棒石负载有机胺吸附剂的制备
1.4 酸改性凹凸棒石及负载有机胺吸附剂的表征
1.5 酸改性凹凸棒石负载有机胺吸附剂的性能评价
2 结果与讨论
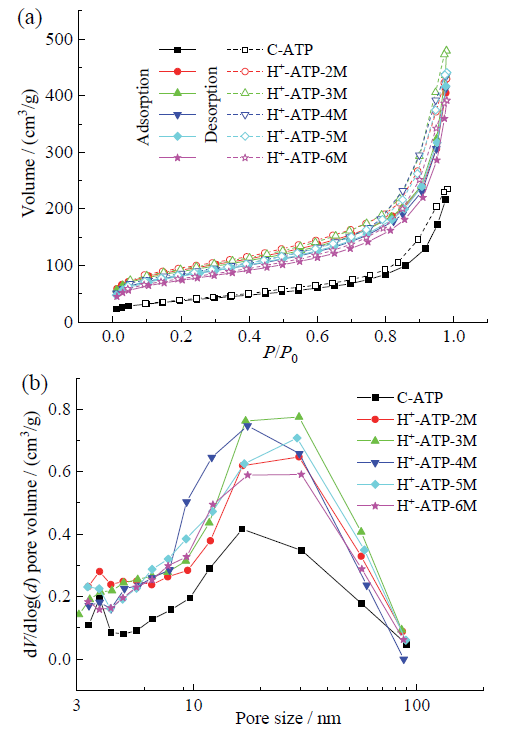
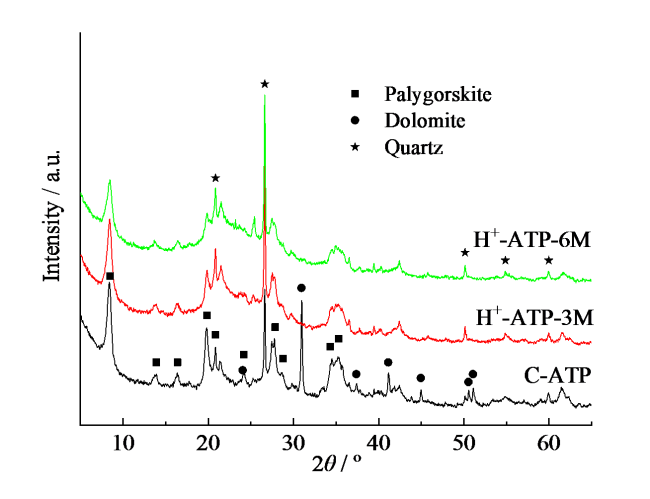
2.1 酸改性浓度对凹凸棒石组成、形貌、结构和性能的影响
Table 1 Chemical composition of crude attapulgite and acid-activated attapulgite by different acid concentration表1 凹凸棒石原矿和不同酸浓度改性凹凸棒石的化学组成 |
| Sample | Oxide content / % | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | Fe2O3 | MgO | CaO | K2O | TiO2 | MnO | P2O5 | Na2O | |
| C-ATP | 60.75 | 13.40 | 8.86 | 9.85 | 3.86 | 1.74 | 1.13 | 0.16 | 0.13 | 0.12 |
| H+-ATP-2M | 78.75 | 8.52 | 4.59 | 5.41 | 0.25 | 1.05 | 1.36 | 0.04 | 0.03 | 0.10 |
| H+-ATP-3M | 83.28 | 6.25 | 3.30 | 4.38 | 0.37 | 0.78 | 1.53 | 0.02 | 0.03 | 0.08 |
| H+-ATP-4M | 83.54 | 5.74 | 3.02 | 4.05 | 0.20 | 0.73 | 1.49 | 0.02 | 0.02 | 0.07 |
| H+-ATP-5M | 84.62 | 5.31 | 2.85 | 3.61 | 0.17 | 0.72 | 1.53 | 0.03 | 0.03 | 0.08 |
| H+-ATP-6M | 85.83 | 5.24 | 2.60 | 3.48 | 0.16 | 0.72 | 1.52 | 0.01 | 0.02 | 0.08 |
Fig. 1 N2 adsorption-desorption isotherms (a) and BJH pore-size distribution (b) of crude attapulgite and acid-activated attapulgite by different acid concentration图1 凹凸棒石原矿和不同酸浓度改性凹凸棒石的N2吸附-脱附等温线(a)和BJH孔径分布图(b) |
Table 2 Structural parameters of crude attapulgite and acid-activated attapulgite by different acid concentration表2 凹凸棒石原矿和不同酸浓度改性凹凸棒石的结构参数 |
| Sample | Surface area / (m2/g) | Pore volume / (cm3/g) | Dp / nm | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SBET | Smicro | Sext | Vtotal | Vmicro | Vmeso | Vmacro | ||||
| C-ATP | 145.3 | 15.7 | 129.6 | 0.413 | 0.007 | 0.386 | 0.020 | 8.1 | ||
| H+-ATP-2M | 313.4 | 81.9 | 231.5 | 0.665 | 0.041 | 0.574 | 0.050 | 8.5 | ||
| H+-ATP-3M | 304.9 | 80.4 | 224.5 | 0.741 | 0.040 | 0.667 | 0.034 | 9.7 | ||
| H+-ATP-4M | 281.8 | 67.2 | 214.6 | 0.673 | 0.033 | 0.605 | 0.035 | 9.6 | ||
| H+-ATP-5M | 267.2 | 58.4 | 208.8 | 0.642 | 0.025 | 0.583 | 0.034 | 9.7 | ||
| H+-ATP-6M | 255.7 | 50.8 | 204.9 | 0.606 | 0.025 | 0.542 | 0.039 | 9.5 | ||
Fig. 2 SEM images of crude attapulgite and acid-activated attapulgite by different acid concentration: (a) C-ATP; (b) H+-ATP-3M; (c) H+-ATP-6M图2 凹凸棒石原矿和不同酸浓度改性凹凸棒石的SEM图像:(a)C-ATP;(b)H+-ATP-3M;(c)H+-ATP-6M |
Fig. 3 XRD patterns of crude attapulgite and acid-activated attapulgite by different acid concentration图3 凹凸棒石原矿和不同酸浓度改性凹凸棒石的XRD谱图 |
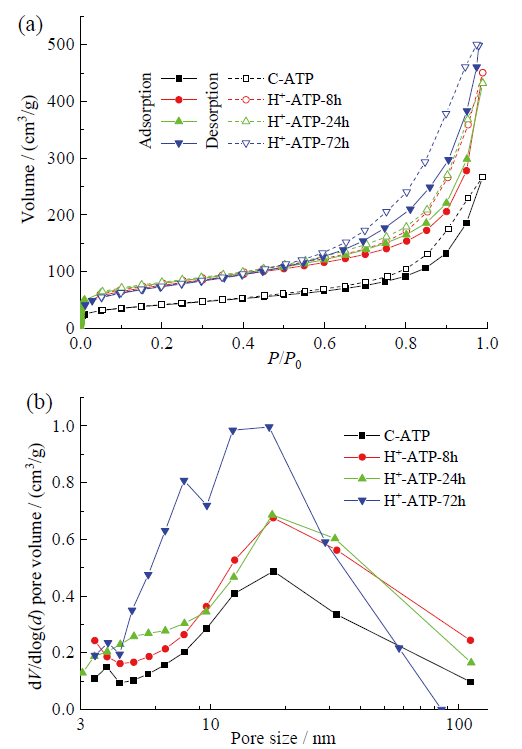
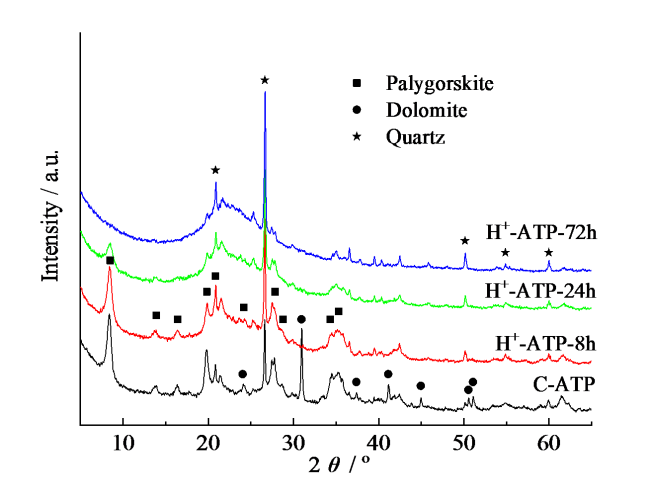
2.2 酸改性时间对凹凸棒石组成、形貌、结构和性能的影响
Table 3 Chemical composition of crude attapulgite and acid-activated attapulgite by different time表3 凹凸棒石原矿和不同时间酸改性凹凸棒石的化学组成 |
| Sample | Oxide content / % | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | Fe2O3 | MgO | CaO | K2O | TiO2 | MnO | P2O5 | Na2O | |
| C-ATP | 60.75 | 13.40 | 8.86 | 9.85 | 3.86 | 1.74 | 1.13 | 0.16 | 0.13 | 0.12 |
| H+-ATP-8h | 81.76 | 7.00 | 3.59 | 4.73 | 0.34 | 0.92 | 1.51 | 0.02 | 0.03 | 0.12 |
| H+-ATP-24h | 89.78 | 4.03 | 1.70 | 2.01 | 0.18 | 0.74 | 1.43 | 0.01 | 0.02 | 0.10 |
| H+-ATP-72h | 93.87 | 2.73 | 0.74 | 0.43 | 0.12 | 0.72 | 1.29 | 0.01 | 0.02 | 0.09 |
Fig. 4 N2 adsorption-desorption isotherms (a) and BJH pore-size distribution (b) of crude attapulgite and acid-activated attapulgite by different time图4 凹凸棒石原矿和不同时间酸改性凹凸棒石的N2吸附-脱附等温线(a)和BJH孔径分布图(b) |
Table 4 Structural parameters of crude attapulgite and acid-activated attapulgite by different time表4 凹凸棒石原矿和不同时间酸改性凹凸棒石的结构参数 |
| Sample | Surface area / (m2/g) | Pore volume / (cm3/g) | Dp / nm | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SBET | Smicro | Sext | Vtotal | Vmicro | Vmeso | Vmacro | ||||
| C-ATP | 145.3 | 15.7 | 129.6 | 0.413 | 0.007 | 0.386 | 0.020 | 8.1 | ||
| H+-ATP-8h | 265.1 | 48.5 | 216.6 | 0.699 | 0.024 | 0.628 | 0.047 | 10.5 | ||
| H+-ATP-24h | 278.0 | 67.5 | 210.5 | 0.669 | 0.031 | 0.609 | 0.029 | 9.6 | ||
| H+-ATP-72h | 260.9 | 4.3 | 256.6 | 0.773 | 0.001 | 0.767 | 0.005 | 11.9 | ||
Fig. 5 SEM images of crude attapulgite and acid-activated attapulgite by different time: (a) C-ATP; (b) H+-ATP-8h; (c) H+-ATP-24h; (d) H+-ATP-72h图5 凹凸棒石原矿和不同时间酸改性凹凸棒石的SEM图:(a)C-ATP;(b)H+-ATP-8h;(c)H+-ATP-24h;(d)H+-ATP-72h |
Fig. 6 XRD patterns of crude attapulgite and acid-activated attapulgite by different time图6 凹凸棒石原矿和不同时间酸改性凹凸棒石的XRD谱图 |
2.3 有机胺种类对CO2吸附性能的影响
Table 5 CO2 adsorption capacity of adsorbents prepared by different organic amines supported on α-SiO2表5 α-SiO2负载不同有机胺所制备吸附剂的CO2吸附量 |
| Types of organic amines | CO2 adsorption capacity / (mmol/g) |
|---|---|
| MEA | 2.14 |
| DEA | 2.66 |
| TEA | 2.70 |
| DETA | 2.81 |
| TETA | 2.85 |
| PEI | 3.21 |
Table 6 Comparison of CO2 adsorption capacity of PEI-modified adsorbents from literatures and this study表6 PEI改性不同载体制备的吸附剂的CO2吸附量的比较 |
| Support | Capacity / % | Temperature / oC | CO2 adsorption capacity / (mmol/g) | References |
|---|---|---|---|---|
| SBA-15 | 50 | 25 | 1.87 | [23] |
| Palygorskite | 25 | 25 | 0.89 | [8] |
| Montmorillonite | 50 | 75 | 2.55 | [12] |
| Al2O3 | 60 | 50 | 1.87 | [24] |
| Carbon black | 50 | 75 | 3.5 | [25] |
| PMMA | 10 | 40 | 2.4 | [26] |
| α-SiO2 | 50 | 60 | 3.21 | This study |