0 引言
1 实验部分
1.1 SiOCN材料合成
1.2 电极材料制备及极片预锂化
1.3 材料表征
1.4 电化学性能测试
2 结果与讨论
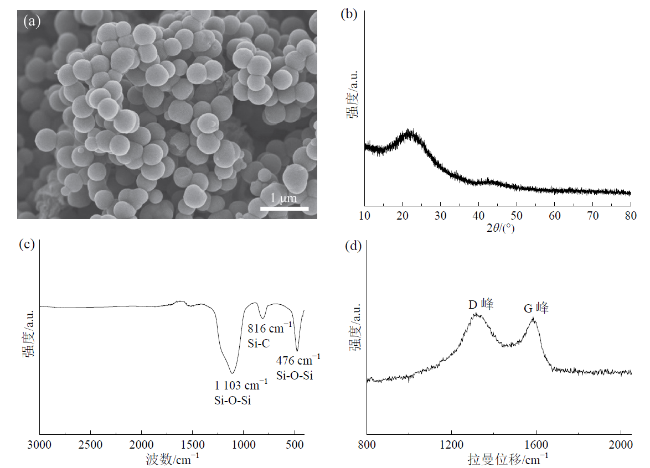
Fig. 1 SEM image (a), XRD pattern (b), FTIR spectra (c), and Raman spectra (d) of SiOCN material图1 SiOCN材料的SEM图像(a)、XRD图谱(b)、FTIR谱图(c)和拉曼谱图(d) |
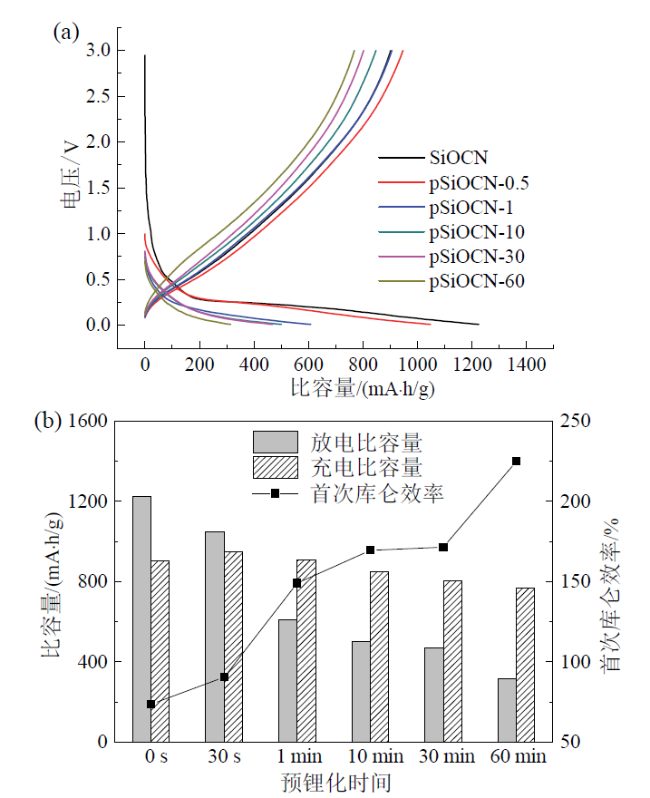
Fig. 2 Initial charge/discharge curves (a), and initial charge/discharge capacities and Coulombic efficiency (b) of SiOCN electrodes with different prelithiation time图2 不同预锂化时间SiOCN电极的首次充放电曲线(a)及首次充放电比容量和首次库仑效率(b) |
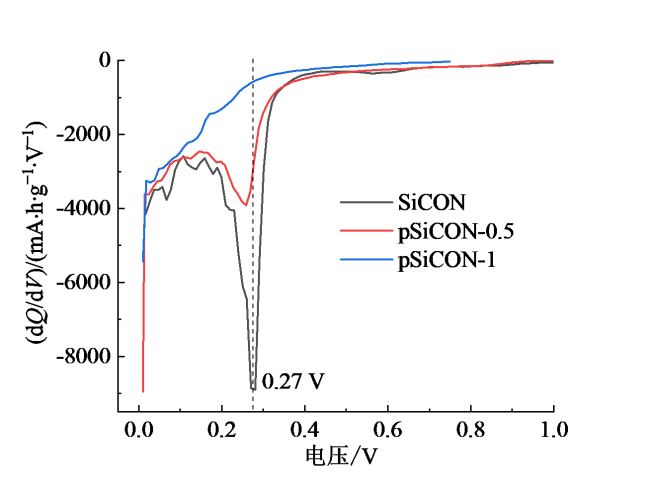
Fig. 3 The dQ/dV curves derived from the discharge profiles of SiOCN, pSiOCN-0.5 and pSiOCN-1 electrodes图3 SiOCN、pSiOCN-0.5和pSiOCN-1电极放电曲线的dQ/dV曲线 |
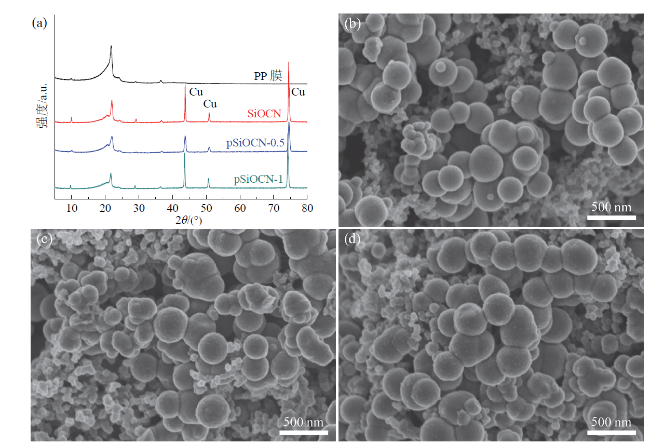
Fig. 4 XRD pattern of SiOCN electrodes (a); SEM images of SiOCN (b), pSiOCN-0.5 (c), and pSiOCN-1 (d)图4 SiOCN电极的XRD图谱(a);SiOCN(b)、pSiOCN-0.5(c)、pSiOCN-1(d)的SEM图 |
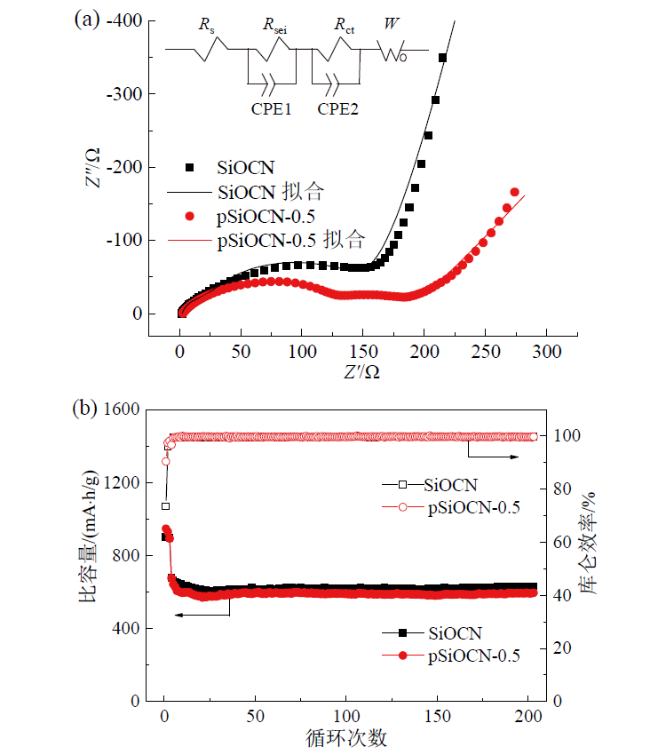
Fig. 5 EIS spectra before cycling (a) and cycling performance at 1 A/g (b) of SiOCN and pSiOCN-0.5 electrodes图5 SiOCN和pSiOCN-0.5电极循环前的EIS谱(a)及1 A/g循环性能(b) |
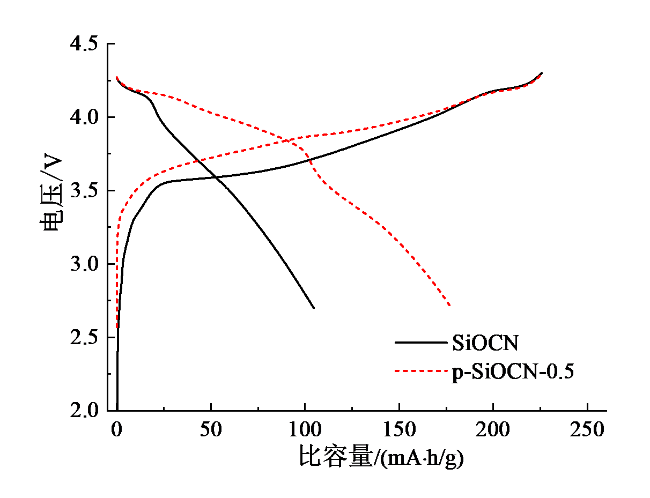
Fig. 6 Initial charge/discharge curves of NCM811/SiOCN and NCM811/pSiOCN-0.5 full cells图6 NCM811/SiOCN和NCM811/pSiOCN-0.5全电池的首次充放电曲线 |